单选题
下列说法中正确的是( )
- A、 “冰,水为之,而寒于水”说明相同质量的冰和水比较,冰的能量更高
- B、 “蜡炬成灰泪始干”中“泪”的主要成分是水
- C、 “南朝四百八十寺,多少楼台烟雨中”的“烟雨”是由飘浮在空气中的固体小颗粒形成的
- D、 “钻石恒久远,一颗永流传”说明常温下钻石的化学性质比较稳定
已知:△G=△H-T△S,当△G<0,反应能自发进行,△G>0反应不能自发进行。下列说法中正确的是( )
- A、 非自发反应在任何条件下都不能发生
- B、 熵增加且放热的反应一定是自发反应
- C、 自发反应一定是熵增加的反应,非自发反应一定是熵减小或不变的反应
- D、 凡是放热反应都是能自发进行的反应,而吸热反应都是非自发进行的反应
下列装置中,都伴随有能量变化,其中是由化学能转变为电能的是( )
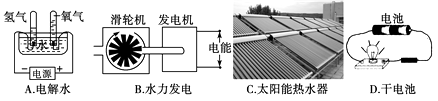
- A、 A
- B、 B
- C、 C
- D、 D
一定条件下,在水溶液中1molCl-、ClOx-(x=1,2,3,4)的能量(kJ)相对大小如图所示。下列有关说法正确的是( )
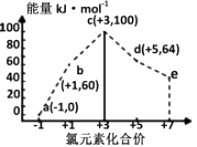
- A、 a、b、c、d、e中,c最稳定
- B、 b→a+c反应的活化能为反应物能量减生成物能量
- C、 b→a+d反应的热化学方程式为:3ClO-(aq)=ClO3-(aq)+2Cl-(aq)ΔH=+116kJ·mol-1
- D、 一定温度下,Cl2与NaOH溶液反应生成的产物有a、b、d,溶液中a、b、d的浓度之比可能为11∶1∶2
已知: ①C6H12O6(s)=2C2H5OH(l)+2CO2(g) △H1
②6CO2(g)+6H2O(g)=C6H12O6(s)+6O2 △H2
③2H2O(g)=2H2(g)+O2(g) △H3
④2CO2(g)+6H2(g)=C2H5OH(l)+3H2O △H4
下列有关说法正确的是( )
- A、 H2的燃烧热为
- B、 反应①使用催化剂,△H1将减小
- C、 标准状况下,反应②生成1.12LO2 , 转移的电子数为0.1×6.02×1023
- D、 2△H4=△H1+△H2-6△H3
已知C2H2 (g) + O2 (g) → 2CO2 (g) + H2O (g)+1256 kJ,下列说法正确的是( )
- A、 1 份乙炔完全燃烧可放热1256 kJ
- B、 反应中乙炔做还原剂、被还原
- C、 乙炔完全燃烧时,生成2 mol液态水,所放热量小于2512 kJ
- D、 若有10 mol电子发生转移,则需消耗2.5 mol O2
下列变化过程中Q>0的是( )
- A、 CH3COOH
CH3COO- + H+ + Q
- B、 CaCO3(s)→ CaO(s)+ CO2(g)+ Q
- C、 S2- + H2O
HS- + OH- + Q
- D、 S(g)→S(l)+ Q
对H2在O2中燃烧的反应,下列说法不正确的是 ( )
- A、 该反应为吸热反应
- B、 该反应为放热反应
- C、 断裂H—H键吸收能量
- D、 生成H—O键放出能量
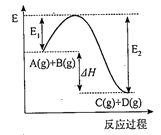
某反应的反应过程中能量变化如图所示(图中E1表示正反应的活化能,E2表示逆反应的活化能).下列有关叙述正确的是( )

- A、 该反应为放热反应
- B、 催化剂能改变反应的焓变
- C、 逆反应的活化能大于正反应的活化能
- D、 催化剂能降低反应的活化能
已知H+(aq)+OH﹣(aq)=H2O(l)△H=﹣57.3kJ/mol,向50mL 2mol/L的NaOH溶液中加入1mol/L的某种酸恰好完全反应,测得加入酸的体积与反应放出热量的关系如下图所示(不考虑热量的散失),则该酸可能是( )

- A、 醋酸(CH3COOH)
- B、 盐酸
- C、 草酸(HOOC﹣COOH)
- D、 硫酸
通过以下反应均可获取O2。下列有关说法不正确的是 ( )
①光催化分解水制氧气:2H2O(l)=2H2(g) + O2(g) ΔH1=+571.6 kJ·mol-1
②过氧化氢分解制氧气:2H2O2(l)=2H2O(l) + O2(g) ΔH2=-196.4 kJ·mol-1
③一氧化氮分解制氧气:2NO(g)=N2(g) + O2(g) ΔH3=-180.5 kJ·mol-1
- A、 反应①是人们获取H2的途径之一
- B、 反应②、③都是化学能转化为热能
- C、 反应H2O2(l)=H2O(g)+
O2(g)的ΔH=-285.8kJ·mol-1
- D、 反应2H2(g)+2NO(g)=N2(g)+2H2O(l)的ΔH=-752.1 kJ·mol-1
参照反应Br+H2→HBr+H的能量随反应过程变化的示意图(如下图所示),下列叙述中正确的是( )
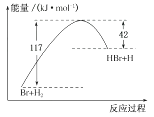
- A、 反应物具有的总能量大于生成物具有的总能量
- B、 正反应为吸热反应
- C、 该反应的逆反应是吸热过程
- D、 从图中可以看出,HBr的能量一定高于H2的能量
如图为反应2H2(g)+O2(g)═2H2O(g)的能量变化示意图.下列说法正确的是( )
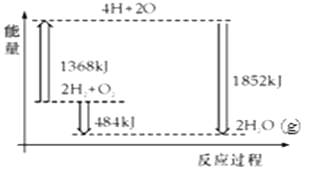
- A、 H2(g)和O2(g)反应生成H2O(g),这是吸热反应
- B、 2molH2和1molO2转化为4molH、2molO原子的过程中放出热量
- C、 2molH2(g)和1molO2(g)反应生成2molH2O(g),共放出484kJ能量
- D、 4molH、2molO生成2molH2O(g),共放出484kJ能量