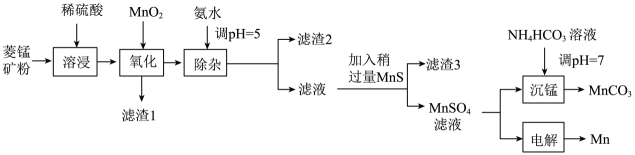
单选题
化学与社会、生产、生活密切相关。下列说法错误的是
- A、 久置的红薯变甜,是因为葡萄糖发生了水解
- B、 电热水器用镁棒防止金属内胆腐蚀,原理是牺牲阳极保护法
- C、 第五形态的碳单质“碳纳米泡沫”,与石墨烯互为同素异形体
- D、 用太阳能光催化分解水代替电解水制氢气有利于节能环保
下列物质应用错误的是
- A、
溶液用作水果的保鲜剂
- B、 苯甲酸及其钠盐用作食品防腐剂
- C、
用作生活用水的消毒剂
- D、 用热NaOH溶液洗去铁表面沾有的柴油
下列实验装置或操作正确的是

- A、 用图1装置测定中和反应的反应热
- B、 用图2装置测定化学反应速率
- C、 用图3装置分离乙醚和苯
- D、 用图4装置振荡萃取、静置分层后,打开分液漏斗上方的玻璃塞再进行分液
- A、 1mol
中含有Si-O键的数目为2
- B、 25℃、101kPa下,4g
中含有的原子数为2
- C、 标准状况下,6.72L
与水充分反应转移的电子数目为0.1
- D、 25℃时,1.0L
的
溶液中含有
的数目为0.2
某种天然沸石的化学式为 , 其中元素X、Y、Z、R、W原子序数依次增大,且占据四个不同周期。Y在地壳中含量最高,在该化合物中R显示其最高价态,基态W原子的核外电子恰好填满10个原子轨道。下列说法错误的是
, 其中元素X、Y、Z、R、W原子序数依次增大,且占据四个不同周期。Y在地壳中含量最高,在该化合物中R显示其最高价态,基态W原子的核外电子恰好填满10个原子轨道。下列说法错误的是
- A、 简单离子半径:
- B、 第一电离能:
- C、 最简单氢化物稳定性:
- D、 氯化物熔点:
下列实验操作能达到实验目的的是
- A、 向溶液中加入硝酸酸化的氯化钡溶液,检验溶液中的
- B、 向沸水中逐滴加入饱和氯化铁溶液并继续加热搅拌制备氢氧化铁胶体
- C、 除去NaCl固体中的少量
, 将固体溶解后蒸发结晶、趁热过滤、洗涤干燥
- D、 将
固体溶解于盛有适量蒸馏水的烧杯中,经转移、洗涤、定容和摇匀,配制一定浓度的
溶液
7-ACCA是合成头孢克罗的关键中间体,其结构如图所示。下列说法正确的是

- A、 该化合物的化学式为
- B、 分子中有5种不同化学环境的氢原子
- C、 分子中含有3个手性碳原子
- D、 不存在分子中含有苯环的同分异构体
pH计的工作原理(如图所示)是通过测定电池电动势E(即玻璃电极和参比电极的电势差)而确定待测溶液的pH。电池电动势E与待测溶液pH关系为: (E的单位为V,K为常数)。下列说法错误的是
(E的单位为V,K为常数)。下列说法错误的是

- A、
计工作时,化学能转化为电能
- B、 玻璃电极玻璃膜内外
的差异会引起电池电动势的变化
- C、 若玻璃电极电势比参比电极低,玻璃电极反应:
- D、 若测得
的标准溶液电池电动势E为
, 可标定常数
实验室初步分离苯甲酸乙酯、苯甲酸和环己烷的流程如下:

已知:苯甲酸乙酯的沸点为212.6℃,“乙醚-环己烷-水共沸物”的沸点为62.1℃。下列说法错误的是
- A、 操作a和操作b不同
- B、 操作c为重结晶
- C、 无水
和饱和碳酸钠溶液的作用相同
- D、 由该流程可以说明苯甲酸和苯甲酸钠在水中的溶解度差别很大
《环境科学》刊发了我国科研部门采用零价铁活化过硫酸钠( , 其中S为+6价)去除废水中正五价砷As(V)的研究成果,其反应机理模型如图所示。下列说法错误的是
, 其中S为+6价)去除废水中正五价砷As(V)的研究成果,其反应机理模型如图所示。下列说法错误的是
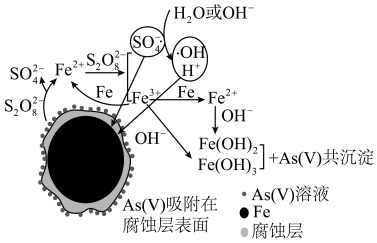
- A、 反应过程中有非极性键的断裂
- B、 溶液的pH越小,越有利于去除废水中的正五价砷
- C、 碱性条件下,硫酸根自由基发生的反应方程式为
- D、 反应过程中,存在反应
我国科学家设计如下图所示“电子转移链”过程,实现了低温条件下氧化 高效制
高效制 。
。

下列说法错误的是
- A、 反应Ⅰ为取代反应
- B、
水解可制取
- C、 参加反应的
 和
和的物质的量之比为2:1
- D、 1mol
 最多能与4mol
最多能与4mol 发生反应