试题详情
金属钨(W)可做白炽灯泡的灯丝。用黑钨矿[主要含有FeWO4(钨酸亚铁)]制得金属钨(W)的工艺流程如下图所示: H2O+WO3 ④3H2+WO3
H2O+WO3 ④3H2+WO3  W+3H2O
W+3H2O
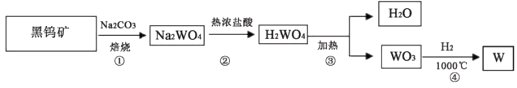
其主要反应原理如下:
①4FeWO4+4Na2CO3+O2 4Na2WO4+2Fe2O3+4CO2
②Na2WO4+2HCl(热浓)=H2WO4+2NaCl
③H2WO4下列说法错误的是( )
- A、反应③属于分解反应
- B、反应①②③④的产物都有氧化物
- C、反应①④中有元素的化合价发生改变
- D、为加快反应的速率,可将黑钨矿石碾成粉末
知识点

参考答案
采纳过本试题的试卷